FOSSIL FUELS: ENVIRONMENTAL EFFECTS
It is perhaps ironic that the same forces which are responsible for producing pollution allow us to eliminate it. With industrial development comes prosperity. Industrial development is accompanied, and indeed is made possible, by the exponential growth in energy consumption, which in turn has resulted in air pollution. And prosperity allows us to begin to think about cleaning up the 'mess' that we have made. (Most underdeveloped nations cannot afford to clean up their increasingly polluted environments.)
There are two main issues that we need to discuss here:
- conventional pollution (acid rain, smog, etc.)
- greenhouse effect
The former is a major problem now, especially in the industrialized nations and large cities; we know how to solve this problem and there is increasing political determination to solve it. The latter is a controversial issue whose impact may be seen in the 21st century; there are no easy ways to solve this problem, despite some initial efforts.
Elementary chemistry is sufficient to evaluate the contribution of different fossil fuels to carbon dioxide emissions. Let's take the simplest case of natural gas. It is essentially pure methane, CH4. Its heating value is about 24,000 BTU per pound. Its combustion can be represented as folows:
CH4 + O2 = CO2 + 2H2O
So for every 16 pounds (or tons) of methane that are consumed, 12 pounds (or tons) of carbon are released (as carbon dioxide) and 44 pounds (or tons) of carbon dioxide are produced. (Remember that the atomic weights of carbon, oxygen and hydrogen are 12, 16 and 1, respectively.) In 1993, out of 84.0 quads of energy consumed in the U.S., 19.0 came from burning natural gas, as shown in our general discussion of fossil fuels. Therefore, carbon emissions from natural gas combustion are obtained as follows:

This is about 20% of the total quantity of carbon emitted (270/1350) by the U.S., as illustrated below (upper curve). (Also shown are carbon emissions of the other G-7 countries (lower curves), as reported by the Energy Information Administration.) Note that this is less than the 26.3% contribution of natural gas to total fossil fuel combustion (19.0/(19.0+32.7+20.5)=26.3%).

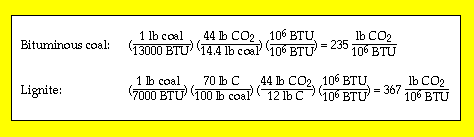
Coal contributes the most to carbon emissions; the lower the rank, the greater its contribution per unit of energy released. This is illustrated below. Petroleum's contributions are intermediate between those of natural gas and coal. (See also Table 11-3 in the textbook.)
Because the greenhouse effect is a global problem - even the observatory in Hawaii is registering the exponential growth of CO2 emissions - it is important to compare carbon emissions not only among industrialized nations, but also among the currently underdeveloped countries, whose energy consumption (and thus CO2 emissions) in the 21st century is expected to be very rapid. The choice of the most appropriate basis for such comparisons (per capita, per $GDP, etc.) is an important unresolved issue, and it will have a profound effect on international negotiations about emission reductions. The following table summarizes the data for selected countries (from (a) World Almanac 1996 and (b) "Energy Use and Carbon Emissions: Some International Comparisons", EIA, DOE, March 1994).
 Obviously, the rankings of the culprits will be very different when expressed in absolute terms, or per capita, or per dollar of GDP.
Obviously, the rankings of the culprits will be very different when expressed in absolute terms, or per capita, or per dollar of GDP.
For some of the recent media accounts of the greenhouse effect, click here.
For some of the recent media accounts of conventional air pollution, click here.
This clip from The New York Times is a good introduction to our discussion of conventional air pollution.

Similar pollution index measurements are being reported increasingly by the media, and are of increasing concern to city dwellers. For example, there is a recent report (see The Daily Collegian of 10/10/95) about a link between carbon monoxide pollution in cities and hospital admissions for heart failure among the elderly. There is also the widely publicized case of excess mortality in London in December 1952 attributed to high levels of SO2 and smoke.
lrr3@psu.edu (last revised 12/6/95)





